Chapter 1 Properties of Gases
-
The properties of gases
gas - the simplest state of matter (fills any container it
occupies)
- perfect gas can be
pictured as a collection of molecules in continuous random motion, with speeds
that increase with Temp >. Different
than a liq. in that, except during collisions, the molecules of the gas are
widely separated from one another and move in paths that are largely unaffected by intermolecular
forces.
The physical state of a substance is defined by its physical properties,
and two samples of a substance that have the same physical properties are in
the same state.
The state of a pure gas is specified by giving values of V,
n, P, & T.
p = f (T,V,n) general form of equation of state
Experimentally determination of 3 of the 4 properties fixes the
fourth and the properties of a simple system.
f (P, V, T, n ) = 0(All 4 variables are not independent of
each other)
Mathematical form for the perfect or ideal gas:
R is the gas constant.
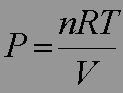
The perfect gas obeys the perfect gas equation (mathematical expression)
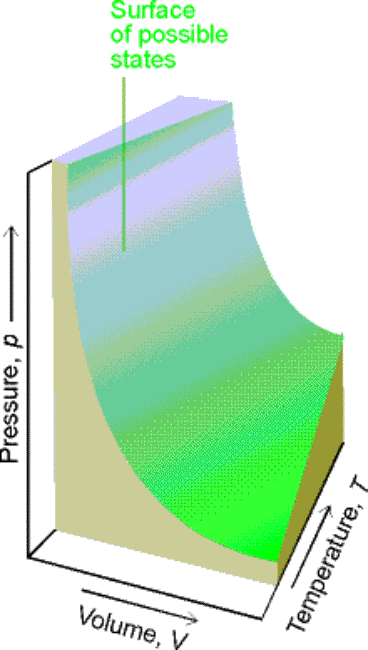
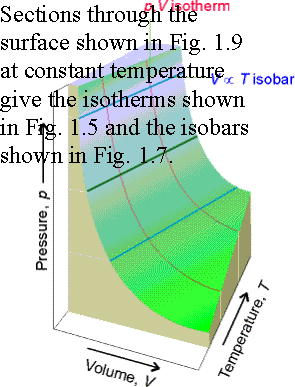
Fig. 1.9
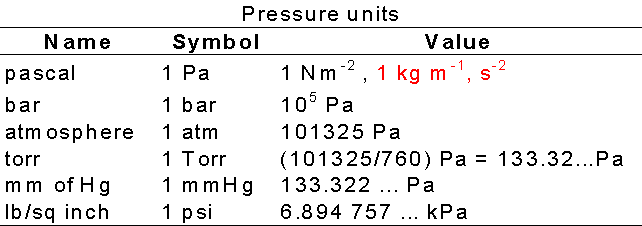
Pressure is defined as force divided by the area to which the force
is applied (force per unit area). Normally reported in Pascals, Pa, where 1 Pa
= 1 N m-2 , & 1 atm = 1.01325 x 105 Pa.
A pressure of 105 Pa (1 bar) is the standard
pressure for reporting data, denoted Pø.
Calculating Pressure – Example
Dibutyl phthalate is commonly used as a liquid in manometers.
What pressure is equivalent to a centimeter of this liquid?
r for dibutyl phthalate is = 1.0465 g/cc, g
= 9.8067 m/s2
P = f/A = mg/A = mgh/V = rgh
f=ma V=hxA r =m/V
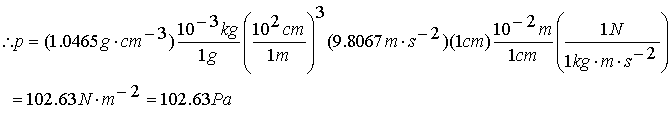
Calculate the pressure exerted by a mass of 1 kg pressing through
the point of a pin of area 1 x 10-2 mm2 @ Earth’s
surface?
P = F/A = mg/A =1kg (9.81m/s2)/(.01mm2)(1000mm/1m)2
P = 980,000,000 Pa = 9.7 x 103
atm.
Pressure exerted by a column of liquid is given by:
P = rgh , (F = mg = rAhg)
Measurement of pressure of a gas inside a container can be done
with a manometer.
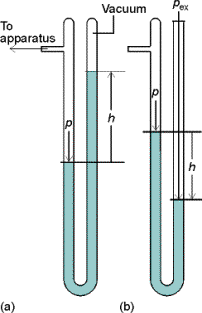
Fig.
1.2 a) P = rgh b) P = Pex + rgh
Temperature
The concept of temperature springs from the observation that a change in physical state (ie V) can occur when two objects are in contact with one another.
Temperature is a property that tells us whether two objects would
be in thermal equilibrium if they were in contact through a diathermic
boundary. A boundary is Diathermic
if a change of state is observed when two objects of different temperatures are
brought into contact.
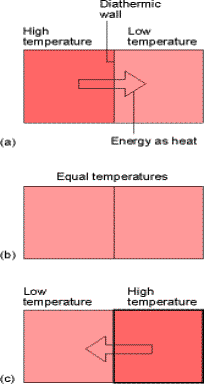
An adiabatic
wall is a boundary where no change occurs between two objects with
different temperature. (insulating
walls)
Fig. 1.3
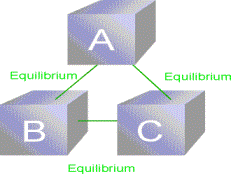
The Zeroth Law of Thermodynamics: If A is in
thermal equilibrium with B, and B is in thermal equilibrium with C, then C
is also in thermal equilibrium with A.
The Zeroth law justifies the concept of temperature and the use of
a thermometer. (e.g. mercury
thermometer)
Early experiments with liquid thermometers arbitrarily divided the freezing point and boiling point of water into 100 units to give the Celcius temperature scale of 0 °C. for ice and 100 °C for boiling water at 1 atm pressure. Due to use of many liquids/materials in the thermometer, different expansions occurred resulting in different scales.
The pressure of a gas can be used to construct a perfect-gas
temperature scale (virtually independent of gas). This is also identical to
the thermodynamic temperature scale.
Temperature on the thermodynamic temperature scale is reported in
kelvins, K.
T/K = q/°C + 273.15 (definition of Celsius temp. in terms of kelvin
temp.)
Gas Laws
Boyle 1661 showed PV = constant - Boyle’s Law
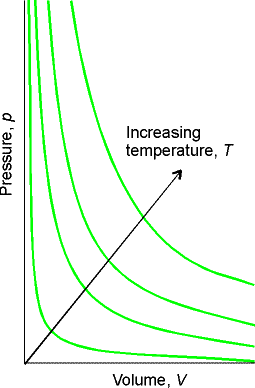
Fig. 1.5

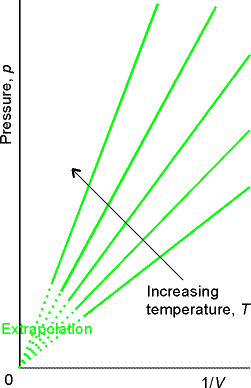

Fig. 1.6
Molecular interpretation of Boyle’s Law:
As a sample is compressed to half its volume, twice as many
molecules strike the walls in a given period of time than before
compression. Results in doubling
average force on the walls.
Valid only at low pressures (limiting law).
Next important addition came from Jacques Charles:
Volume increases linearly with temperature independent of the
identity of the gas.
Charles Law: V = constant x T (
at constant pressure)

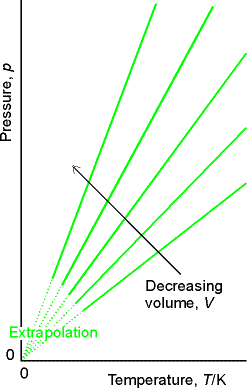
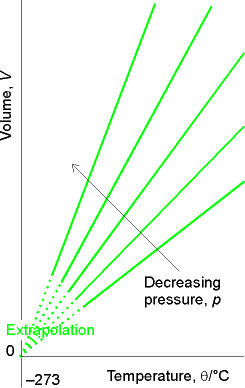

Fig.
1.7 Fig. 1.8
Molecular explanation of Charles’s Law:
Raising temperature of a gas increases the average speed of its molecules. The molecules collide with the wall more frequently and with greater impact, thus exerting greater pressure.
Final piece of data: At a
given pressure and temperature, the molar volume, Vm = V/n,
volume per mole of molecules, of a gas is the same regardless of the identity
of the gas.
V = constant x n (at constant T & P), the constant of
proportionality is independent of the identity of the gas and only depends upon
the number of molecules.
Avogadro’s Principle: equal volumes of gas at same T&P have
the same number of molecules.
Combining the three contributions gives the equation of state for
a perfect gas PV = nRT, where R is denoted as the gas constant. This equation is approximate for any gas and
becomes increasingly exact as the pressure of the gas goes to zero.
Gas constant R is determined experimentally in the limit of
pressure tending to zero. R = 8.31451 J
K-1
mol-1 =
8.20578 x 10-2 L atm K-1 mol-1 =
8.31451 x 10-2 L bar K-1 mol-1

Application of the Perfect Gas Equation
A commercial gas cylinder contains 75 L of Helium at 15 bar (gauge pressure). Assuming ideal gas behavior for the isothermal expansion, how many 3.0 L balloons at a pressure of 1.1 bar can be filled by the gas in the cylinder?
Method - Summarize known and unknown data as follows:

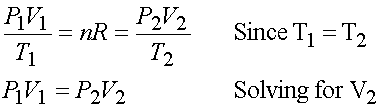
V2 = V1 (P1/P2) = 75 L (16 bar/ 1.1 bar) = 1100 liters
We need to subtract the 75 L left in cylinder to give 1025 L
to fill balloons at 3 liters each. Which finally gives
1025 L/ (3L / balloon) = ~341 balloons.
Mixtures of gases:
Dalton’s Law: The pressure exerted by a mixture of perfect gases is the
sum of the partial pressures of the gases.
The partial pressure of a perfect gas is the pressure it would
exert if it occupied the container alone.
The total Pressure P = PA + PB + PC + .... (sum of partial pressures) with each substance J, PJ = nJRT/V
Example of using Partial pressures:
A 1 gram sample of liquid water is added to 1 L of N2 gas at 1.01 bar
and 25 0C. To what volume
must the system be adjusted in order for all the water to evaporate? Assume
ideal gas behavior. What will be the
total pressure of the system? The vapor
pressure of water is 0.0316 bar @ 25 0 C.
All the water will evaporate when the partial pressure of water in the container equals its vapor pressure. The volume of the system must be such that the pressure of water vapor is 0.0316 bar.
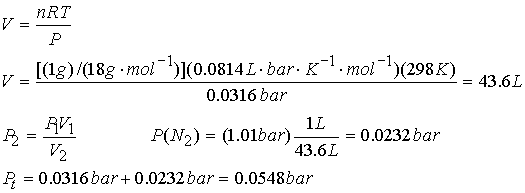
The Mole fraction, xj , is the amount
of J expressed as a fraction of the total amount of molecules, n, in the
sample.
xJ = nJ / n where n = nA + nB + .....
It follows that xA + xB + .... = 1
We define Partial Pressure pJ , of a gas J in
a mixture ( perfect or real) as pJ = xJp , where p is
total pressure.
Also pA + pB + ..... = (xA + xB + .... ) p = p
Example 1:
The pressure of a gaseous mixture of NH3 and N2 decreased from
1.5 kPa to 1 kPa after all the NH3 was absorbed from the
mixture. Assume ideal gas behavior and
find initial composition of the mixture.
Partial pressure of N2 is 1 kPa , PT = PNH + PN
P(NH3) = PT - P(N2) = 1.5 kPa - 1
kPa = 0.5 kPa
from Dalton’s Law : x(N2) = P(N2)/PT = 1 kPa/1.5kPa =
0.67
x(NH3) = 1- x(N2) = 0.33